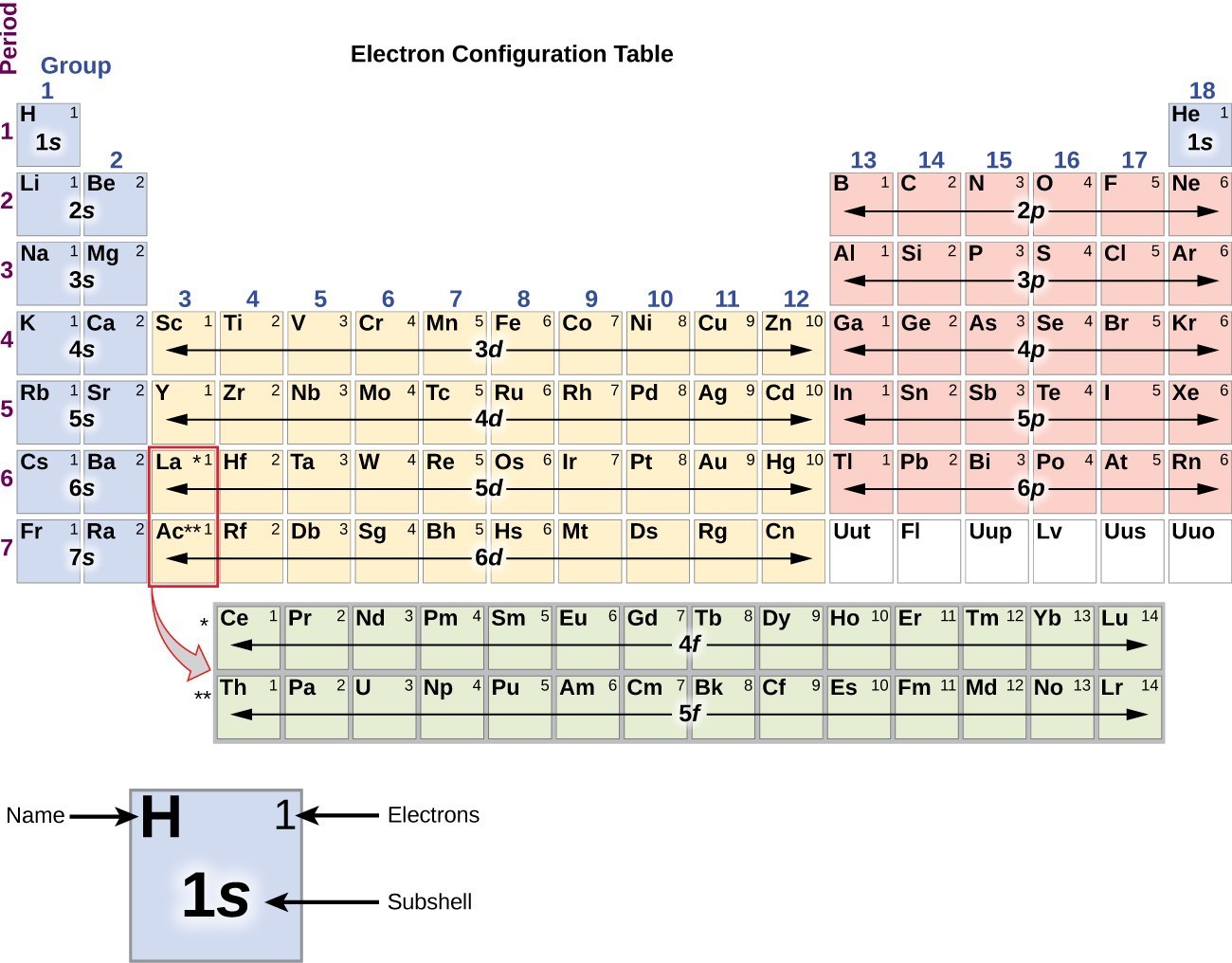
Greater the number of parallel spins of electrons more stable the orbital. An orbital can be occupied only by two electrons having opposite spins. Aufbau principle: Electrons first occupy orbitals with lower energy than higher energy. Thus, the obtained lower energy configuration is known as ground-state electron configuration. Concept introduction: By using Aufbau principle, electrons starts to occupy from orbitals of lower energy level. The ground-state electron configuration of selenium ( Se ) has to be written using Aufbau principle. Write the ground-state electron configuration:Īfter filling all the orbitals according to the Aufbau principle, the resulting electron configuration is the ground-state electron configuration of Selenium. 
In the case of Selenium, you would first fill the 4s orbital with 2 electrons, followed by the 3d orbital with 10 electrons, and finally the 4p orbital with 4 electrons. Starting from the lowest energy level, fill the orbitals with the appropriate number of electrons. This means that the 4s orbital has the lowest energy, followed by the 3d orbital, and finally the 4p orbital. The energy of the orbitals can be ranked as follows: 4 s < 3 d < 4 p. Compare the energy levels of the orbitals: In the case of Selenium, the orbitals with lower energy are 4s, 3d, and 4p. 
The electron configuration of Selenium is 4 s 2 3 d 10 4 p 4.Īccording to the Aufbau principle, electrons first occupy orbitals with lower energy before filling the ones with higher energy. That is two electrons in an atom can have same three quantum number ( n, l, m l ) but the spin quantum number differs because of electrons in same orbital spins in opposite direction. The order of electrons filling in an atom is given as Figure 1 Pauli Exclusion Principle: It states that in an atom no two electrons have same quantum numbers. Each electron fills each orbital till it is half filled. Selenium has 34 electrons, so its electron configuration is: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p4 In this configuration, the first shell has 2 electrons, the second shell has 8 electrons, the third shell has 18 electrons, and the fourth shell has 6 electrons.Question asked by Filo student The ground-state electron configuration of selenium ( Se ) has to be written using Aufbau principle. Now that we understand the basics, let’s look at the full electron configuration of selenium. Therefore, the electron configuration of the third shell is 3s2 3p6 3d10. The 3s orbital can hold 2 electrons, while each of the 3 p orbitals can hold 2 electrons. The electrons fill the 3s, 3p, and 3d orbitals. The third shell, or the M shell, can hold a maximum of 18 electrons. Therefore, the electron configuration of the second shell is 2s2 2p6. The 2s orbital can hold 2 electrons, while each of the 3 p orbitals can hold 2 electrons. The electrons fill the 2s and 2p orbitals. The second shell, or the L shell, can hold a maximum of 8 electrons.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
